GIRBL Gruppe
Group: Immune Cell Migration & Vascular Biology
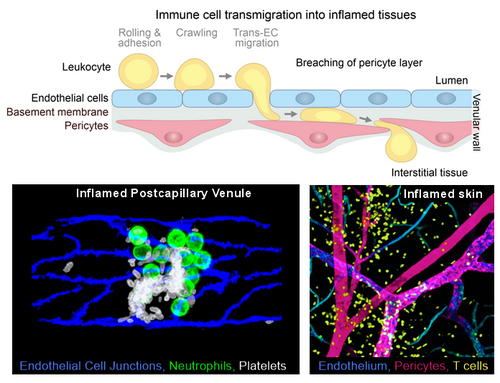
Immune cell recruitment from the circulation into tissues is essential for host defense but also a major driver of inflammatory disease. During this process, leukocytes cross the venular wall - a multicellular barrier composed of endothelial cells, pericytes, and the basement membrane.
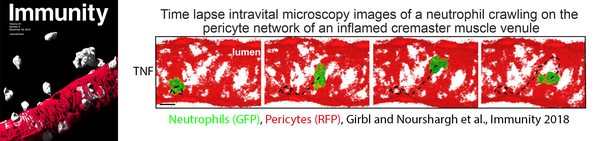
While the endothelial step of this process is well understood, our research focuses on the underexplored post-endothelial phase, where immune cells navigate within the venular wall before entering inflamed tissues. Our work has established that distinct, spatially organized chemokine cues direct neutrophil transmigration across the different layers of venular walls and that neutrophil post-endothelial crawling on pericytes is essential for efficient tissue entry. We aim to uncover how this intramural microenvironment controls immune cell entry into inflamed tissues.
Research goals
Our general goal is to define the cellular and molecular mechanisms that govern leukocyte trafficking across venular walls in vivo. We focus on how neutrophils are guided through the pericyte layer and how local signals shape their migratory behavior and function. In parallel, we investigate how effector T cells interact with vascular wall components during peripheral inflammation.
By combining advanced confocal and two-photon intravital microscopy with genetic models and quantitative analysis, we dissect immune cell behavior in its native tissue context with high spatial and temporal resolution.
Hackenbroch C, Cimen T, Gross C, Rubenzucker S, Burkard P, Groß N, Ahrends R, Girbl T*, Stegner D*. Neutrophil recruitment depends on platelet-derived leukotriene B4. Blood Advances. 2025 May 13;9(9):2226-2230. doi: 10.1182/bloodadvances.2024014947. *Corresponding authors.
Joulia R, Guerrero-Fonseca IM, Girbl T, Coates JA, Stein M, Vázquez-Martínez L, Lynam E, Whiteford J, Schnoor M, Voehringer D, Roers A, Nourshargh S, Voisin MB. Neutrophil breaching of the blood vessel pericyte layer during diapedesis requires mast cell-derived IL-17A. Nat Commun. 2022 Nov 17;13(1):7029. doi: 10.1038/s41467-022-34695-7.
Barkaway A, Rolas L, Joulia R, Bodkin J, Lenn T, Owen-Woods C, Reglero-Real N, Stein M, Vázquez-Martínez L, Girbl T, Poston RN, Golding M, Saleeb RS, Thiriot A, von Andrian UH, Duchene J, Voisin MB, Bishop CL, Voehringer D, Roers A, Rot A, Lämmermann T, Nourshargh S. Age-related changes in the local milieu of inflamed tissues cause aberrant neutrophil trafficking and subsequent remote organ damage. Immunity. 2021 Jul 13;54(7):1494-1510.e7. doi: 10.1016/j.immuni.2021.04.025.
Owen-Woods C, Joulia R, Barkaway A, Rolas L, Ma B, Nottebaum AF, Arkill KP, Stein M, Girbl T, Golding M, Bates DO, Vestweber D, Voisin MB, Nourshargh S. Local microvascular leakage promotes trafficking of activated neutrophils to remote organs. J Clin Invest 2020; p133661, doi: 10.1172/JCI133661.
Girbl T, Lenn T, Perez L, Rolas L, Barkaway A, Thiriot A, Del Fresno C, Lynam E, Hub E, Thelen M, Graham G, Alon R, Sancho D, von Andrian UH, Voisin MB, Rot A, Nourshargh S. Distinct Compartmentalization of the Chemokines CXCL1 and CXCL2 and the Atypical Receptor ACKR1 Determine Discrete Stages of Neutrophil Diapedesis. Immunity 2018;49(6):p1062-1076, doi: 10.1016/j.immuni.2018.09.018.
Gutjahr JC, Szenes E, Tschech L, Asslaber D, Schlederer M, Roos S, Yu X, Girbl T, Sternberg C, Egle A, Aberger F, Alon R, Kenner L, Greil R, Orian-Rousseau V, Hartmann TN. Microenvironment-induced CD44v6 promotes early disease progression in chronic lymphocytic leukemia. Blood 2018;22;131(12):p1337-1349, doi: 10.1182/blood-2017-08-802462.
Schulz-Fincke AC, Tikhomirov AS, Braune A, Girbl T, Gilberg E, Bajorath J, Blaut M, Nourshargh S, Gütschow M. Design of an Activity-Based Probe for Human Neutrophil Elastase: Implementation of the Lossen Rearrangement To Induce Förster Resonance Energy Transfers. Biochemistry 2018;57(5):p742-752, doi: 10.1021/acs.biochem.7b00906.
Ganghammer S, Hutterer E, Hinterseer E, Brachtl G, Asslaber D, Krenn PW, Girbl T, Berghammer P, Geisberger R, Egle A, Zucchetto A, Kruschinski A, Gattei V, Chigaev A, Greil R, Hartmann TN. CXCL12-induced VLA-4 activation is impaired in trisomy 12 chronic lymphocytic leukemia cells: a role for CCL21. Oncotarget 2015;20;6(14):p12048-60, doi: 10.18632/oncotarget.3660.
Girbl T, Lunzer V, Namberger K, Greil R, Hartmann TN. The CXCR4 and adhesion molecule expression of CD34+ hematopoietic cells mobilized by on-demand addition of plerixafor to G-CSF. Transfusion 2014;54(9):p 2325-35, doi: 10.1111/trf.12632.
Asslaber D, Grossinger EM, Girbl T, Hofbauer SW, Egle A, Weiss L, Greil R, Hartmann TN. Mimicking the microenvironment in chronic lymphocytic leukaemia - where does the journey go? Br J Haematol 2013;160:p 711-714, doi: 10.1111/bjh.12151.
Girbl T, Hinterseer E, Grossinger EM, Asslaber D, Oberascher K, Weiss L, Hauser-Kronberger C, Neureiter D, Kerschbaum H, Naor D, Alon R, Greil R, Hartmann TN. CD40-mediated activation of chronic lymphocytic leukemia cells promotes their CD44-dependent adhesion to hyaluronan and restricts CCL21-induced motility. Cancer Research 2013;73:p 561-570, doi: 10.1158/0008-5472
Brachtl G, Sahakyan K, Denk U, Girbl T, Alinger B, Hofbauer SW, Neureiter D, Hofbauer JP, Egle A, Greil R, Hartmann TN. Differential bone marrow homing capacity of VLA-4 and CD38 high expressing chronic lymphocytic leukemia cells. PLoS One 2011;6:p e23758, doi: 10.1371/journal.pone.0023758.
Gadermaier G, Hauser M, Egger M, Ferrara R, Briza P, Santos KS, Zennaro D, Girbl T, Zuidmeer-Jongejan L, Mari A, Ferreira F. Sensitization prevalence, antibody cross-reactivity and immunogenic peptide profile of Api g 2, the non-specific lipid transfer protein 1 of celery. PLoS One 2011;6:p e24150, doi: 10.1371/journal.pone.0024150.
Gadermaier G, Egger M, Girbl T, Erler A, Harrer A, Vejvar E, Liso M, Richter K, Zuidmeer L, Mari A, Ferreira F. Molecular characterization of Api g 2, a novel allergenic member of the lipidtransfer protein 1 family from celery stalks. Mol Nutr Food Res 2011;55:p 568-577, doi: 10.1002/mnfr.201000443.
Gadermaier G, Harrer A, Girbl T, Palazzo P, Himly M, Vogel L, Briza P, Mari A, Ferreira F. Isoform identification and characterization of Art v 3, the lipid-transfer protein of mugwort pollen. Mol Immunol 2009;46:p 1919-1924, doi: 10.1016/j.molimm.2009.03.021.
Dr. Tamara Girbl
Josef-Schneider-Str. 2

Tugce Cimen
Josef-Schneider-Str. 2

Dr. Marilia Fernandes Manchope
Josef-Schneider-Str. 2

Luisa Klinkenberg
Josef-Schneider-Str. 2
Current position
Group leader at the Rudolf Virchow Center of the University of Wuerzburg (since June 2020)
Research Experience
2018 – 2020 Postdoctoral Researcher with Prof. Michael Sixt, Institute of Science and Technology (IST) Austria
2014 – 2018 Postdoctoral Fellow of the British Heart Foundation with Prof. Sussan Nourshargh, Queen Mary University of London, United Kingdom
2009 – 2012 PhD with Prof. Tanja Hartmann and Prof. Richard Greil, University Clinics Salzburg, Austria
Current position
Group leader at the Rudolf Virchow Center of the University of Wuerzburg (since June 2020)
Research Experience
2018 – 2020 Postdoctoral Researcher with Prof. Michael Sixt, Institute of Science and Technology (IST) Austria
2014 – 2018 Postdoctoral Fellow of the British Heart Foundation with Prof. Sussan Nourshargh, Queen Mary University of London, United Kingdom
2009 – 2012 PhD with Prof. Tanja Hartmann and Prof. Richard Greil, University Clinics Salzburg, Austria
Education
2013 PhD (Dr. rer. nat.) in Genetics and Molecular Biology, University of Salzburg, Austria
Funding, Fellowships and Awards
- 2025 Project Leader (PI), DFG Research Training Group RTG 3190
- 2021 Start-up grant from the University of Würzburg
- 2020 PRIMA Fellowship, Swiss National Science Foundation (offer gratefully declined)
- 2019 Servier Award for Microcirculation
- 2017 Young Investigator Award of the European Society for Microcirculation
- 2016 Travel & presentation awards from the Gordon Research Conference on Chemotactic Cytokines
- 2016 Travel fellowship, European Chemokine and Cell Migration Meeting
- 2014 – 2018 BHF Immediate Postdoctoral Fellowship
- 2014 – 2016 Marie Curie Co-Fund Postdoctoral Fellowship
- 2013 Sanofi publication award
- 2013 Wilhelm Türk publication award from the Austrian Society for Hematology & Oncology
- 2009 – 2013 International PhD Programme Immunity in Cancer and Allergy (FWF, Austria)
Professional Service
Elected Executive Board member of the Rudolf Virchow Center, University of Wuerzburg (since 2024)