Molecular Microscopy of Polyneuropathy
- in collaboration with PD Kathrin Doppler
Polyneuropathy (PNP) describes a spectrum of diseases involving damage to the peripheral nervous system that affects 5-8% of the population. Recently, a new clinical-histological classification of PNP, paranodopathy, has been described, with primary damage to the nodes of Ranvier in myelinated nerves. We believe that interrogating the protein ring architecture of these nerves is key for proper classification of PNP and integral for therapeutic decision and prognosis. This requires multi-color super-resolution imaging of human tissue. Previously, we investigated human sural nerve biopsies but because these are hard to justify from a clinical standpoint which makes sample acquisition from healthy controls impossible, we want to move to skin biopsies. Since the protein architecture of nodes of Ranvier shows a ring-like structure with a periodicity of ~190 nm, we explore the linear periodicity of neurofascin, a structural protein of the node of Ranvier. Our first results show that “direct Stochastik Optical Reconstruction Microscopy” (dSTORM) imaging of nodes in human skin biopsies is technically possible, and promising for fine diagnostics on human nerve biopsies. Here, the imaging pipeline is complex for skin biopsies as the nerve fiber density is realtively low, while the signal background and optical aberrations are relatively high in tissue. Thus, the immunostaining has to be optimized for dSTORM and tissue slices, and the imaging routine automated to find all nerve fibers of interest to be imaged and analyzed.
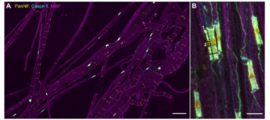
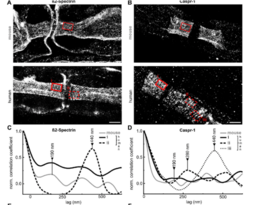
Figure: High-content (left A,B, scale bar 50 µm and 5 µm) and super-resolution (right, A,B, scale bar 2 µm) imaging to analyse axonal loss and disruption of the ultrastructural periodic arrangements of paranodal proteins in patients with polyneuropathies (derived from Appeltshauser et al. 2023).
Publications:
Super-resolution imaging pinpoints the periodic ultrastructure at the human node of Ranvier and its disruption in patients with polyneuropathy. Appeltshauser L, Linke J, Heil HS, Karus C, Schenk J, Hemmen K, Sommer C, Doppler K*, Heinze KG*. Neurobiol Dis. 2023 Jun 15;182:106139. doi: 10.1016/j.nbd.2023.106139. *corresponding author
Anti-pan-neurofascin antibodies induce subclass-related complement activation and nodo-paranodal damage. Appeltshauser L, Junghof H, Messinger J, Linke J, Haarmann A, Ayzenberg I, Baka P, Dorst J, Fisse AL, Grüter T, Hauschildt V, Jörk A, Leypoldt F, Mäurer M, Meinl E, Michels S, Motte J, Pitarokoili K, Stettner M, Villmann C, Weihrauch M, Welte GS, Zerr I, Heinze KG, Sommer C, Doppler K.
Brain. 2023 May 2;146(5):1932-1949. doi: 10.1093/brain/awac418.PMID: 36346134

![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/_processed_/8/4/csm_1111_img_webrvz_718_163_11_6a587cb92c.jpg)