P5 - Consequences of microtubule posttranslational modifications on the physics of the parasite Trypanosoma brucei
Consequences of microtubule posttranslational modifications on the physics of the parasite Trypanosoma brucei
The subpellicular microtubule-based cytoskeleton of Trypanosoma brucei is, in comparison to the highly dynamic microtubules of higher eukaryotes, extremely stable and its main purpose appears to provide structural integrity to the parasite. How this is achieved is largely unknown.
In addition to the presence of microtubule-associated proteins important determinants of microtubule (MT) function are a multitude of post-translational modifications of the microtubule filament (MT-PTMs). In T. brucei, acetylation, polyglutamylation and reversible tyrosination have been described as PTMs, but their functional significance remains unknown. In other systems it has been demonstrated that MT-PTMs regulate mechanical properties of microtubules, flagellar functions (Intraflagellar transport (IFT), motility) and MT-based transport. In mammals, MT-PTMs have been linked to various pathological conditions (e.g. neurodegeneration, infertility).
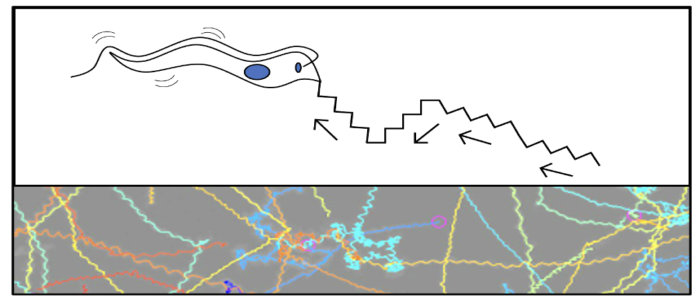
Using genetic engineering methods, such as RNA interference, CRISPR/Cas9 and classical gene knockout methods, enzymes that catalyse such PTMs can be easily manipulated. Using RNAi triggered knockdown of two polyglutamylases, we could show in a previous study that depletion of these enzymes in T. brucei leads to morphological and cytokinesis defects. Since the motility of the parasite is essential, it is of particular interest to know what influence PTMs of tubulin have on the swimming behavior of the cells. For this purpose, additional to the classical cell biological characterisation of the modified cells, we have also developed biophysical methods that make it possible to analyse the motility of wild-type cells and cells with altered PTMs. Here, the focus is mainly on the analysis of the swimming behavior of the cells and the beating frequency of the flagellum. This is performed both in unrestricted chambers to further observe the general swimming pattern, and since trypanosomes must penetrate the vascular system of different host organisms, we would like to investigate the locomotion of individual cells by quantitative tracking experiments (wild type vs. strains with modified microtubule PTMs) in well-controlled microfluidic chambers.