The RNA route to manipulation of the microbiota
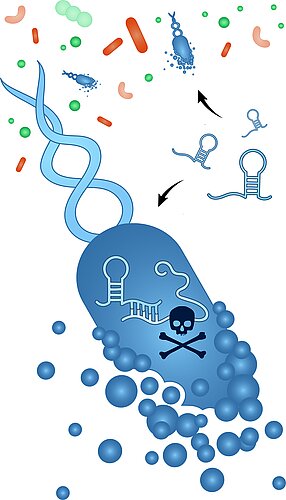
Our body is colonized by a vast array of bacteria the sum of which forms the human microbiome. The gut alone harbors more than 1.000 bacterial species. An understanding of their individual or synergistic contributions to human health and disease demands means to interfere with their functions on the species level. The currently available antibiotics, however, are generally too unspecific for a selective depletion of a single species of interest from the microbiome.
Our ultimate goal is a precision manipulation of microbiota with "programmable antibiotics" as provided by RNA-directed antisense oligonucleotides (ASOs). The same approach can be taken to target antibiotic-resistant pathogen as an alternative to standard antibiotics. These ASOs are coupled to small peptides that carry them inside the bacteria to silence mRNAs of essential genes. There is already proof-of-principle for this exciting concept, but many open questions remain. What does it take to make these ASOs truly species-specific; how can we minimize potential off-targeting; what are the most effective peptides for delivery; what about the hast response and bacterial resistance mechanisms?
Since there is unlikely a one-fits-all solution for all microbiome species, this project can only succeed by integrating diverse expertise from RNA biology, microbiology, immunology, single-cell biology, and pharmacy. lt is also highly interconnected with the basic questions asked in the other research areas in the Vogel lab.