Project Area C
Boron-Modified Biomolecules
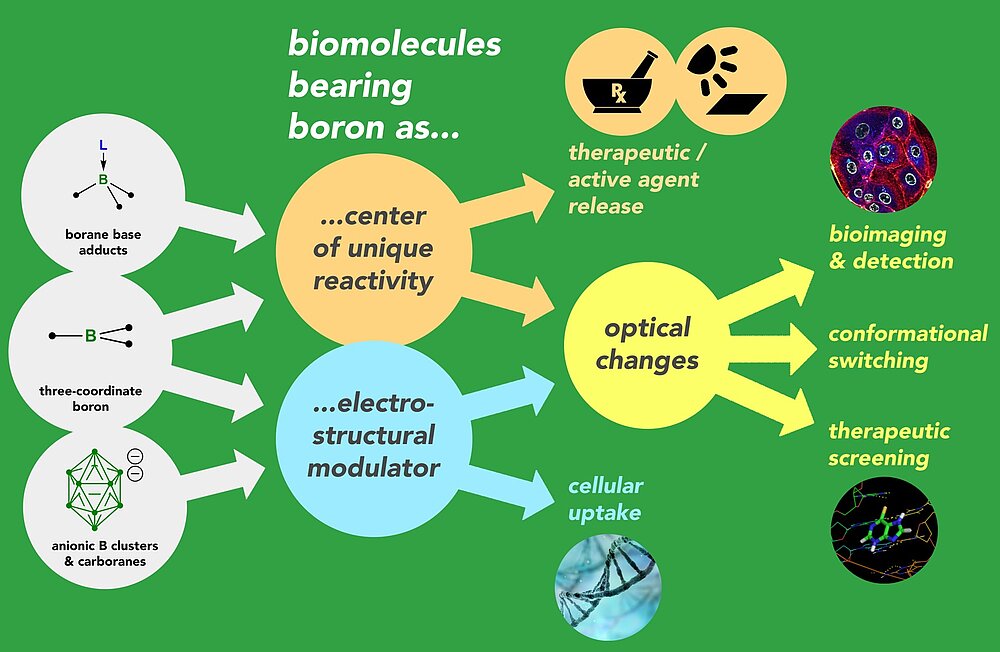
The long-term goal is to use boron’s structural and electronic flexibility to enrich the biochemical toolbox for diagnostic and therapeutic applications.
C01
Project C01 “Boron-Nitrogen Heterocycles as Fluorescent Labels for Nucleic Acids” aims to explore BN-PAHs as a new class of building blocks for chemically modified nucleosides that are of interest as fluorescent probes of nucleic acid structure and function. The goal is to develop syntheses of novel BN-PAH-derived nucleosides and oligonucleotides and the exploration of their structural, photophysical, and fluorescence properties. Responsive fluorescent probes e.g. for DNA structural changes, DNA/RNA hybridization events, protein binding, potentially in real-time are in the focus.
Project leader
C02
Project C02 “Synthesis and Evaluation of Functionalized Boron Clusters for Bioconjugation and Cellular Delivery” aims at the synthesis of boron-cluster-modified biomolecules with enhanced bioavailability and bioactivity. Synthetic strategies for anionic per- and polyhalogenated superchaotropic boron clusters with functional substituents for covalent binding to biomolecular structures will be developed. Principles that modulate the ability to transport various types of cargo through lipid bilayers will be investigated paving the path e.g. to a future development of nucleoside prodrugs and therapeutic oligonucleotides for gene expression regulation.
Project leader
C03
Project C03 “Boronic Acid Ester Chemistry for pH-Response Polymer Mediated Oligonucleotide Delivery” is focused on exploiting boronic acid ester chemistry for the delivery of therapeutic oligonucleotides by developing nanocarriers that incorporate multiple boronic acid (BA) groups in biodegradable polymer nanogels. The project develops the synthesis of block copolymers functionalized with BA or salicylhydroxamic (SHA), examining their ability to form nanoparticles and encapsulate nucleic acids. It also involves designing nucleic acids modified with multiple BA or SHA groups to optimize stability, release efficiency, and loading capacity.
Project leader
C04
Project C04 “Structural Control of Peptides by Boron Photoswitches” aims to develop light-responsive peptide conjugates using boron-containing photoswitches based on cyclic diazobenzenes and related molecules. These site-specifically integrated switches form reversible B–N bonds and respond to specific wavelengths of light to alternate between isomeric structural states, which in turn induce folding or unfolding of α-helical peptides. The photoswitches will be chemically stapled into peptides to control structure, enhance stability, protease resistance, and cell permeability.
Project leaders
C05
Project C05 “ROS-Sensitive 1,2,3-Diazaborinine Prodrugs for Targeted Chemotherapy” aims to develop a highly selective drug delivery system for targeted chemotherapy, based on the ROS-sensitivity of 1,2,3-diazaborinine-based boron cages. Using the anticancer agent camptothecin (CPT), the strategy combines the ROS-responsive prodrug concept with enzymatic tumor targeting. To enhance selectivity, the boron-caged prodrugs are linked to a modified enzyme that locally produces increased ROS levels.
Project leaders








